Flye manual
===========
Table of Contents
-----------------
- [Quick usage](#quickusage)
- [Examples](#examples)
- [Supported Input Data](#inputdata)
- [Parameter Descriptions](#parameters)
- [Flye output](#output)
- [Repeat graph](#graph)
- [Flye benchmarks](#performance)
- [Algorithm Description](#algorithm)
## Quick usage
```
usage: flye (--pacbio-raw | --pacbio-corr | --nano-raw |
--nano-corr | --subassemblies) file1 [file_2 ...]
--genome-size SIZE --out-dir PATH
[--threads int] [--iterations int] [--min-overlap int]
[--meta] [--plasmids] [--no-trestle] [--debug]
[--version] [--help] [--resume]
Assembly of long and error-prone reads
optional arguments:
-h, --help show this help message and exit
--pacbio-raw path [path ...]
PacBio raw reads
--pacbio-corr path [path ...]
PacBio corrected reads
--nano-raw path [path ...]
ONT raw reads
--nano-corr path [path ...]
ONT corrected reads
--subassemblies path [path ...]
high-quality contigs input
-g size, --genome-size size
estimated genome size (for example, 5m or 2.6g)
-o path, --out-dir path
Output directory
-t int, --threads int
number of parallel threads [1]
-i int, --iterations int
number of polishing iterations [1]
-m int, --min-overlap int
minimum overlap between reads [auto]
--asm-coverage int reduced coverage for initial contig assembly [not set]
--plasmids rescue short unassmebled plasmids
--meta metagenome / uneven coverage mode
--no-trestle skip Trestle stage
--polish-target path run polisher on the target sequence
--resume resume from the last completed stage
--resume-from stage_name
resume from a custom stage
--debug enable debug output
-v, --version show program's version number and exit
```
Input reads can be in FASTA or FASTQ format, uncompressed
or compressed with `.gz`. Currently, raw and corrected reads
from PacBio and ONT are supported. Expected error rates are
<30% for raw and <2% for corrected reads. Additionally, the
`--subassemblies` option performs a consensus assembly of multiple
sets of high-quality contigs. You may specify multiple
files with reads (separated by spaces). Mixing different read
types is not yet supported. The `--meta` option enables the mode
for metagenome/uneven coverage assembly.
You must provide an estimate of the genome size as input,
which is used for solid k-mers selection. Standard size
modifiers are supported (e.g. 5m or 2.6g). In the case
of metagenome assembly, the expected total assembly size
should be provided.
To reduce memory consumption for large genome assemblies,
you can use a subset of the longest reads for initial contig
assembly by specifying `--asm-coverage` option. Typically,
30x coverage is enough to produce good draft contigs.
You can separately run Flye polisher on a target sequence
using `--polish-target` option.
## Examples
You can try Flye assembly on these ready-to-use datasets:
### E. coli P6-C4 PacBio data
The original dataset is available at the
[PacBio website](https://github.com/PacificBiosciences/DevNet/wiki/E.-coli-Bacterial-Assembly).
We coverted the raw `bas.h5` file to the FASTA format for the convenience.
wget https://zenodo.org/record/1172816/files/E.coli_PacBio_40x.fasta
flye --pacbio-raw E.coli_PacBio_40x.fasta --out-dir out_pacbio --genome-size 5m --threads 4
with `5m` being the expected genome size, the threads argument being optional
(you may adjust it for your environment), and `out_pacbio` being the directory
where the assembly results will be placed.
### E. coli Oxford Nanopore Technologies data
The dataset was originally released by the
[Loman lab](http://lab.loman.net/2015/09/24/first-sqk-map-006-experiment/).
wget https://zenodo.org/record/1172816/files/Loman_E.coli_MAP006-1_2D_50x.fasta
flye --nano-raw Loman_E.coli_MAP006-1_2D_50x.fasta --out-dir out_nano --genome-size 5m --threads 4
## Supported Input Data
### PacBio data
Flye was tested on raw PacBio reads (P5C3 and P6C4) with error rate ~15%.
Note that Flye assumes that the input files represent PacBio subreads,
e.g. adaptors and noise are trimmed and multiple passes of the same insertion
sequence are separated. This is typically handled by PacBio instruments/toolchains,
however we saw examples of incorrect third-party raw -> fastq conversions,
which resulted into incorrectly trimmed data. In case Flye is failing to
get reasonable assemblies, make sure that your reads are properly preprocessed.
### Oxford Nanopore data
We performed our benchmarks with raw ONT reads (R7-R9) with error rate ~15%.
Due to the biased error pattern, per-nucleotide accuracy is usually lower for
ONT data than with PacBio data, especially in homopolymer regions.
### Error-corrected reads input
While Flye was designed for assembly of raw reads (and this is the recommended way),
it also supports error-corrected PacBio/ONT reads as input (use the ```corr``` option).
The parameters are optimized for error rates <2%. If you are getting highly
fragmented assembly - most likely error rates in your reads are higher. In this case,
consider to assemble using the raw reads instead.
### Consensus of multiple contig sets
```--subassemblies``` input mode generates a consensus of multiple high quality contig assemblies
(such as produced by different short/long read assemblers). The expected error rate
is <1%. You might want to skip the polishing stage with ```--iterations 0``` argument
(however, it might still be helpful to correct small structural errors).
### Input data preparation
Flye works directly with base-called raw reads and does not require any
prior error correction. Flye automatically detects chimeric reads or reads with low quality ends,
so you do not need to curate them before the assembly. However, it is always
worth checking for possible contamination in the reads, since it may affect the
automatic selection of estimated parameters for solid kmers and genome size / coverage.
## Parameter descriptions
### Estimated genome size (required)
You must provide an estimate of the genome size as input,
which is used for solid k-mers selection. The estimate could
be rough (e.g. withing 0.5x-2x range) and does not affect
the other assembly stages. Standard size modificators are
supported (e.g. 5m or 2.6g)
### Minimum overlap length
This sets a minimum overlap length for two reads to be considered overlapping.
In the latest Flye versions, this parameter is chosen automatically
based on the read length distribution (reads N90) and does not require manual setting.
Typical value is 3k-5k (and down to 1k for datasets with shorter read length).
Intuitively, we want to set this parameter as high as possible, so the
repeat graph is less tangled. However, higher values might lead to assembly gaps.
In some *rare* cases (for example in case of biased read length distribution)
it makes sense to set this parameter manualy.
### Metagenome mode
Metagenome assembly mode, that is designed for highly non-uniform coverage and
is sensitive to underrepresented sequence at low coverage (as low as 2x).
In some examples of simple metagenomes, we observed that the normal (isolate)
Flye mode assembled more contigious bacterial
consensus sequence, while the metagenome mode was slightly more fragmented, but
revealed strain mixtures. For relatively complex metagenome `--meta` mode
is the recommended way.
### Reduced contig assembly coverage
Typically, assemblies of large genomes at high coverage require
a hundreds of RAM. For high coverage assemblies, you can reduce memory usage
by using only a subset of longest reads for initial contig extension
stage (usually, the memory bottleneck). The parameter `--asm-coverage`
specifies the target coverage of the longest reads. For a typicall assembly, 30x
is enough to produce good initial contigs. Regardless of this parameter,
all reads will be used at the later pipeline stages.
### Number of polishing iterations
Polishing is performed as the final assembly stage. By default, Flye runs one polishing
iteration. Additional iterations might correct a small number of extra
errors (due to improvements on how reads may align to the corrected assembly).
If the parameter is set to 0, the polishing is not performed.
### Re-starting from a particular assembly stage
Use `--resume` to resume a previous run of the assembler that may have terminated
prematurely (using the same output directory).
The assembly will continue from the last previously completed step.
You might also resume from a particular stage with `--resume-from stage_name`,
where `stage_name` is a choice of `assembly, consensus, repeat, trestle, polishing`.
For example, you might supply different sets of reads for different stages.
## Flye output
The main output files are:
* `assembly.fasta` - Final assembly. Contains contigs and possibly scaffolds (see below).
* `assembly_graph.{gfa|gv}` - Final repeat graph. Note that the edge sequences might be
different (shorter) than contig sequences, because contigs might include multiple
graph edges (see below).
* `assembly_info.txt` - Extra information about contigs (such as length or coverage).
Each contig is formed by a single unique graph edge. If possible, unique contigs are
extended with the sequence from flanking unresolved repeats on the graph. Thus,
a contig fully contains the corresponding graph edge (with the same id), but might
be longer then this edge. This is somewhat similar to unitig-contig relation
in OLC assemblers. In a rare case when a repetitive graph edge is not covered by
the set of "extended" contigs, it will be also output in the assembly file.
Sometimes it is possible to further order contigs into scaffolds based on the
repeat graph structure. These ordered contigs will be output as a part of scaffold
in the assembly file (with a `scaffold_` prefix). Since it is hard to give a reliable estimate of the
gap size, those gaps are represented with the default 100 Ns. `assembly_info.txt`
file (below) contains additional information about how scaffolds were formed.
Extra information about contigs/scaffolds is output into the `assembly_info.txt` file.
It is a tab-delimited table with the columns as follows:
* Contig/scaffold id
* Length
* Coverage
* Is circular (representing circular sequence, such as bacterial chromosome or plasmid)
* Is repetitive (represents repeated, rather than unique sequence)
* Multiplicity (inferred multiplicity based on coverage)
* Graph path (repeat graph path corresponding to this contig/scaffold).
Scaffold gaps are marked with `??` symbols, and `*` symbol denotes a
terminal graph node.
`scaffolds.fasta` file is a symlink to `assembly.fasta`, which is
retained for the backward compatibility.
## Repeat graph
The Flye algorithms are using repeat graph as a core data structure.
In difference to de Bruijn graphs which require exact k-mer matches,
repeat graphs are built using approximate sequence matches, thus
can tollerate higher noise of SMS reads.
The edges of repeat graph represent genomic sequence, and nodes define
the junctions. All edges are classified into unique and repetitive.
The genome traverses the graph in an unknown way, so as each unique
edge appears exactly once in this traversal. Repeat graphs are useful
for repeat analysis and resolution - which are one of the key
genome assembly challenges.
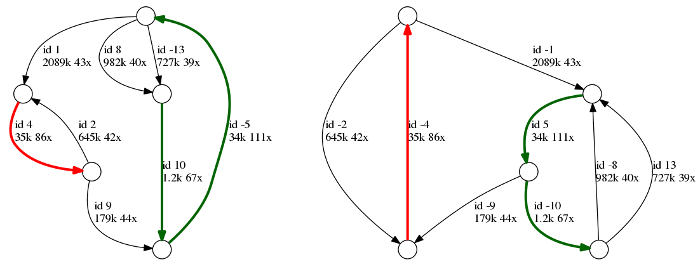
Above is an example of a repeat graph of a bacterial assembly.
Each edge is labeled with its id, length and coverage. Repetitive edges are shown
in color, and unique edges are black. Note that each edge is represented in
two copies: forward and reverse complement (marked with +/- signs),
therefore the entire genome is represented in two copies as well.
In this example, there are two unresolved repeats: (i) a red repeat of
multiplicity two and length 35k and (ii) a green repeat cluster of multiplicity
three and length 34k - 36k. As the repeats remained unresolved, there are no reads
in the dataset that cover those repeats in full. Five unique edges
will correspond to five contigs in the final assembly.
Repeat graphs produced by Flye could be visualized using
[AGB](https://github.com/almiheenko/AGB) or [Bandage](https://github.com/rrwick/Bandage).
Repeat graph before repeat resolution could be found in
the `20-repeat/graph_before_rr.gv` file.
## Flye benchmarks
| Genome | Data | Asm.Size | NG50 | CPU time | RAM |
|--------------------------|----------------|-----------|----------|-----------|--------|
| [E.coli][ecoli] | PB 50x | 4.6 Mb | 4.6 Mb | 2 h | 2 Gb |
| [C.elegans][ce] | PB 40x | 102 Mb | 2.9 Mb | 100 h | 31 Gb |
| [A.thaliana][at] | PB 75x | 120 Mb | 10.7 Mb | 100 h | 46 Gb |
| [D.melanogaster][dm-ont] | ONT 30x | 139 Mb | 17.5 Mb | 130 h | 31 Gb |
| [D.melanogaster][dm-pb] | PB 120x | 142 Mb | 17.5 Mb | 150 h | 75 Gb |
| [Human NA12878][na12878] | ONT 35x (rel6) | 2.9 Gb | 22.6 Mb | 2500 h | 714 Gb |
| [Human CHM13 T2T][t2t] | ONT 50x (rel2) | 2.9 Gb | 57.9 Mb | 3600 h | 871 Gb |
| [Human HG002][hg002] | PB CCS 30x | 2.9 Gb | 30.4 Mb | 1400 h | 272 Gb |
| [Human CHM1][chm1] | PB 100x | 2.8 Gb | 18.8 Mb | 2700 h | 676 Gb |
| [HMP mock][hmp] | PB meta 7 Gb | 66 Mb | 2.6 Mb | 60 h | 72 Gb |
| [Zymo Even][zymo] | ONT meta 14 Gb | 64 Mb | 0.6 Mb | 60 h | 129 Gb |
| [Zymo Log][zymo] | ONT meta 16 Gb | 23 Mb | 1.3 Mb | 100 h | 76 Gb |
[na12878]: https://github.com/nanopore-wgs-consortium/NA12878/blob/master/Genome.md
[ce]: https://github.com/PacificBiosciences/DevNet/wiki/C.-elegans-data-set
[at]: https://downloads.pacbcloud.com/public/SequelData/ArabidopsisDemoData/
[dm-pb]: https://github.com/PacificBiosciences/DevNet/wiki/Drosophila-sequence-and-assembly
[dm-ont]: https://www.ebi.ac.uk/ena/data/view/SRR6702603
[hg002]: https://ftp-trace.ncbi.nlm.nih.gov/giab/ftp/data/AshkenazimTrio/HG002_NA24385_son/PacBio_CCS_15kb/
[ecoli]: https://github.com/PacificBiosciences/DevNet/wiki/E.-coli-Bacterial-Assembly
[hmp]: https://github.com/PacificBiosciences/DevNet/wiki/Human_Microbiome_Project_MockB_Shotgun
[chm1]: https://trace.ncbi.nlm.nih.gov/Traces/sra/?study=SRP044331
[t2t]: https://github.com/nanopore-wgs-consortium/CHM13
[zymo]: https://github.com/LomanLab/mockcommunity
The assemblies generated using Flye 2.5 could be downloaded from [Zenodo](https://zenodo.org/record/3353665).
All datasets were run with default parameters with the following exceptions:
CHM13 T2T was run with `--min-overlap 10000`; CHM1 was run with `--asm-overage 40`;
HG002 was run with maximum read error rate set to 1%.
## Algorithm Description
This is a brief description of the Flye algorithm. Please refer to the manuscript
for more detailed information. The draft contig extension is organized as follows:
* K-mer counting / erroneous k-mer pre-filtering
* Solid k-mer selection (k-mers with sufficient frequency, which are unlikely to be erroneous)
* Contig extension. The algorithm starts from a single read and extends it
with a next overlapping read (overlaps are dynamically detected using the selected
solid k-mers).
Note that we do not attempt to resolve repeats at this stage, thus
the reconstructed contigs might contain misassemblies.
Flye then aligns the reads on these draft contigs using minimap2 and
calls a consensus. Afterwards, Flye performs repeat analysis as follows:
* Repeat graph is constructed from the (possibly misassembled) contigs
* In this graph all repeats longer than minimum overlap are collapsed
* The algorithm resolves repeats using the read information and graph structure
* The unbranching paths in the graph are output as contigs
After resolving bridged repeats, Trestle module attempts to resolve simple unbridged
repeats (of multiplicity 2) using the heterogeneities between repeat copies.
Finally, Flye performs polishing of the resulting assembly
to correct the remaining errors:
* Alignment of all reads to the current assembly using minimap2
* Partition the alignment into mini-alignments (bubbles)
* Error correction of each bubble using a maximum likelihood approach
The polishing steps could be repeated, which might slightly increase quality for some datasets.

